|

Custom Search
|
|
Dermpath-India Pathology of Perineurioma Dr Sampurna Roy MD 2022
|
|
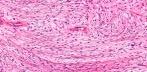
Peripheral nerves are surrounded by an external sheath, which contains concentric layers of thin perineurial cells. Perineuriomas are rare nerve sheath tumours composed of well-differentiated perineurial cells exhibiting immunoreactivity for epithelial membrane antigen (EMA). These lesions occur preferentially in adults and may arise in a wide variety of anatomic sites.
There are three main forms of
perineurioma: This is the common type and presents as a painless subcutaneous mass on the limbs or trunk of middle aged adults. 2. Intraneural Perineurioma: Rare type of lesion. Presents as a swelling of a major nerve in young adults. 3. Sclerosing Perineurioma: Solitary, small nodule in the finger or palm usually in young adults and is associated with deletion of NF2 gene on chromosome 22. Other variants: Fibrous and Reticular Perineuriomas Microscopic features: Well circumscribed non-encapsulated lesion composed of spindle cells with eosinophilic cytoplasm and elongated nuclei.The cells are parallel to each other or form small concentric whorls (onion bulbs). All perineuriomas are EMA positive (focally in some cases), Claudin-1 positive and S100 protein negative. Common electron microscopic features are organelle-poor cell processes, many pinocytotic vesicles, sparse intermediate filaments, tight junctions and patchy external lamina. Sclerosing perineurioma: Well-circumscribed lesion characterized by small epithelioid cells exhibiting corded, trabecular and whorled growth patters together with spindle cells with wavy nuclei end elongated cytoplasmic processes and thick collagen. There is focal whorling of tumour cells. The cells are set in a dense collagenous backround. Immunohistochemically, most of the tumour cells are positive for epithelial membrane antigen, vimentin, collagen type IV and CD10, but not for S-100 protein, CD34, desmin and cytokeratin. The tumor cells are positive for the human erythrocyte glucose transporter (GLUT1) antigen suggesting that this could be an useful marker for the identification of sclerosing perineurioma. Reticular perineurioma: Microscopically the lesions are characterized by a predominantly lace-like or reticular growth pattern composed of anastomosing cords of fusiform cells with bipolar cytoplasmic processes and palely eosinophilic cytoplasm. Nuclei were centrally placed, ovoid to fusiform in shape, and no mitoses were seen. Transition to more cellular areas was focally present in all cases. Differential diagnosis: Myoepithelial tumors, extraskeletal myxoid chondrosarcoma, and myxoid synovial sarcoma. Cutaneous fibrous perineurioma: These tumours presented with sharp circumscription of their deep aspect or were not circumscribed on any side. The cells included plump and spindled with one or more nuclei to thin, elongated, spindled cells with slender nuclei. The backround stroma is fibrotic. Immunohistochemical staining showed EMA-positive staining of the cellular component with collagen type IV-positive staining surrounding the cells. The tumour cells were negative for S-100 protein, factor XIIIa, CD34 and cytokeratin. Perineuriomas may arise in the intestine usually as intramucosal lesions detected as colorectal polyps with distinctive histologic features including entrapment of colonic crypts. Intestinal perineuriomas are distinguished from
other spindle cell neoplasms of the gastrointestinal tract by
immunostaining for EMA and claudin-1. Atypical histologic features (including scattered pleomorphic cells and infiltrative margins) seem to have no clinical significance. Note: Malignant peripheral nerve sheath tumours may show perineurial cell differentiation. These tumours are epithelial membrane antigen positive and S-100 protein negative. The prognosis of perineural MPNST appears to be more favorable than that of conventional MPNST.
|
|
|